Gene editing and gene drives are rapidly emerging as the disruptive technologies du jour. But what are they, what can they do, and why should you care?
Just last week, research was published that took us a step closer to being able to re-engineer whole species by driving specific genes through successive generations – the species in this case was mosquitoes, and the trait to be engineered was the ability to host malaria-causing parasites.
And this week, The U.S. National Academy of Sciences, together with the U.S. National Academy of Medicine, the Chinese Academy of Sciences, and the U.K.’s Royal Society, are co-hosting an international summit on gene editing in humans – and especially the ethical and governance issues emerging capabilities raise.
To help make sense of gene drives and the underlying gene editing technologies, there’s a new explainer video on Risk Bites. Watch the video here, or read the transcript below.
Transcript
Imagine we could stop mosquitoes from carrying malaria. For good. Or prevent ticks from transmitting lyme disease. Or eliminate the billions of dollars of damage caused by bugs to our food supplies each year.
Gene drives are a radical new approach to genetic engineering that could help us achieve these goals, and a whole lot more. Yet, as you might expect, the technology isn’t risk-free.
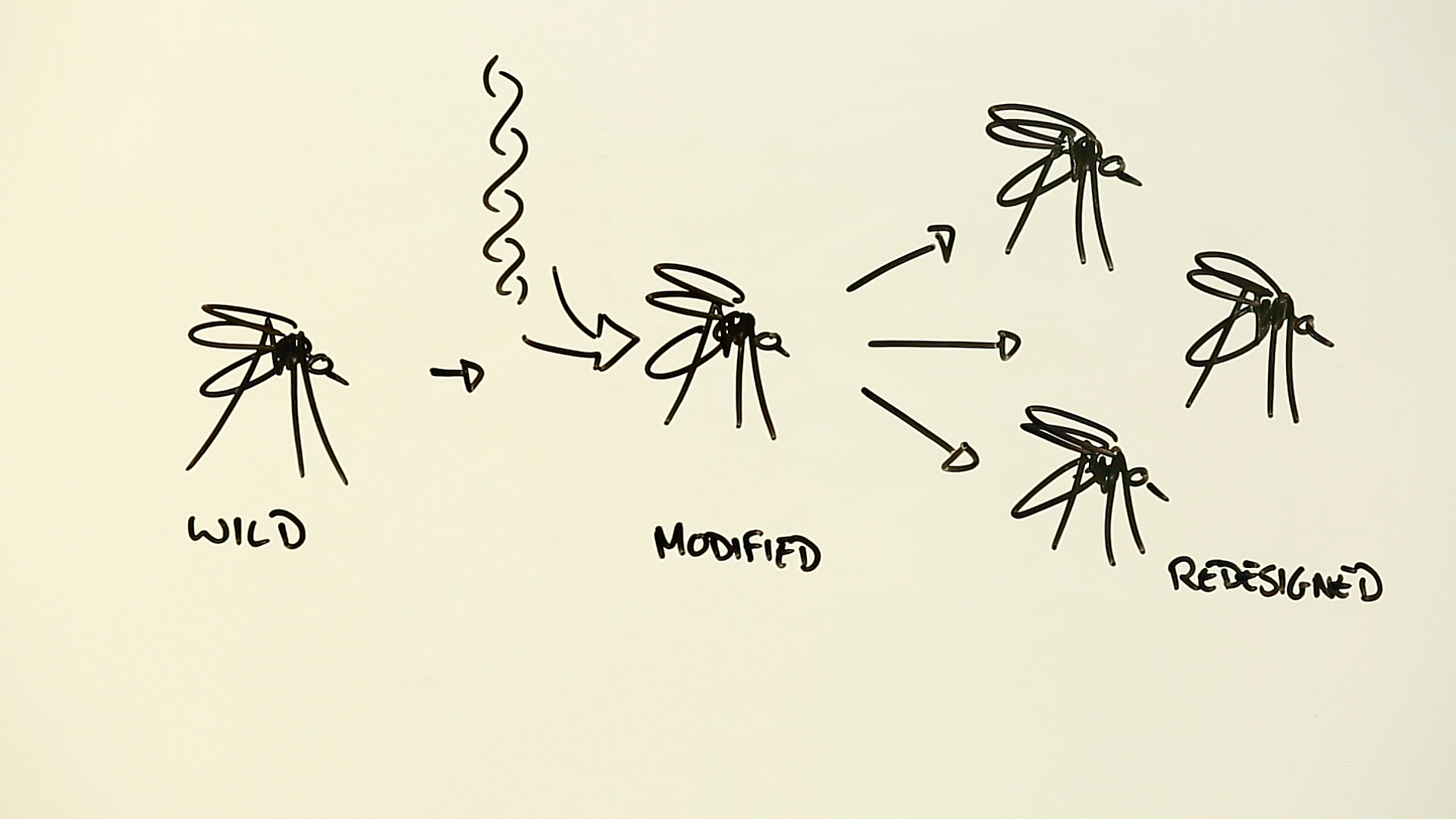
Gene drives are designed to eliminate unwanted traits in insects and other animals. They work by pushing out genetic modifications through whole species, until eventually, every critter has been changed into something we’ve intentionally engineered.
The idea isn’t especially new. But it’s only very recently that advanced gene editing techniques have made human-designed gene drives possible. And at the heart of this revolution is a new technique for precision-editing genes – clustered regularly interspaced short palindromic repeats, or if you’re not into brain-bending tongue twisters, CRISPR for short.
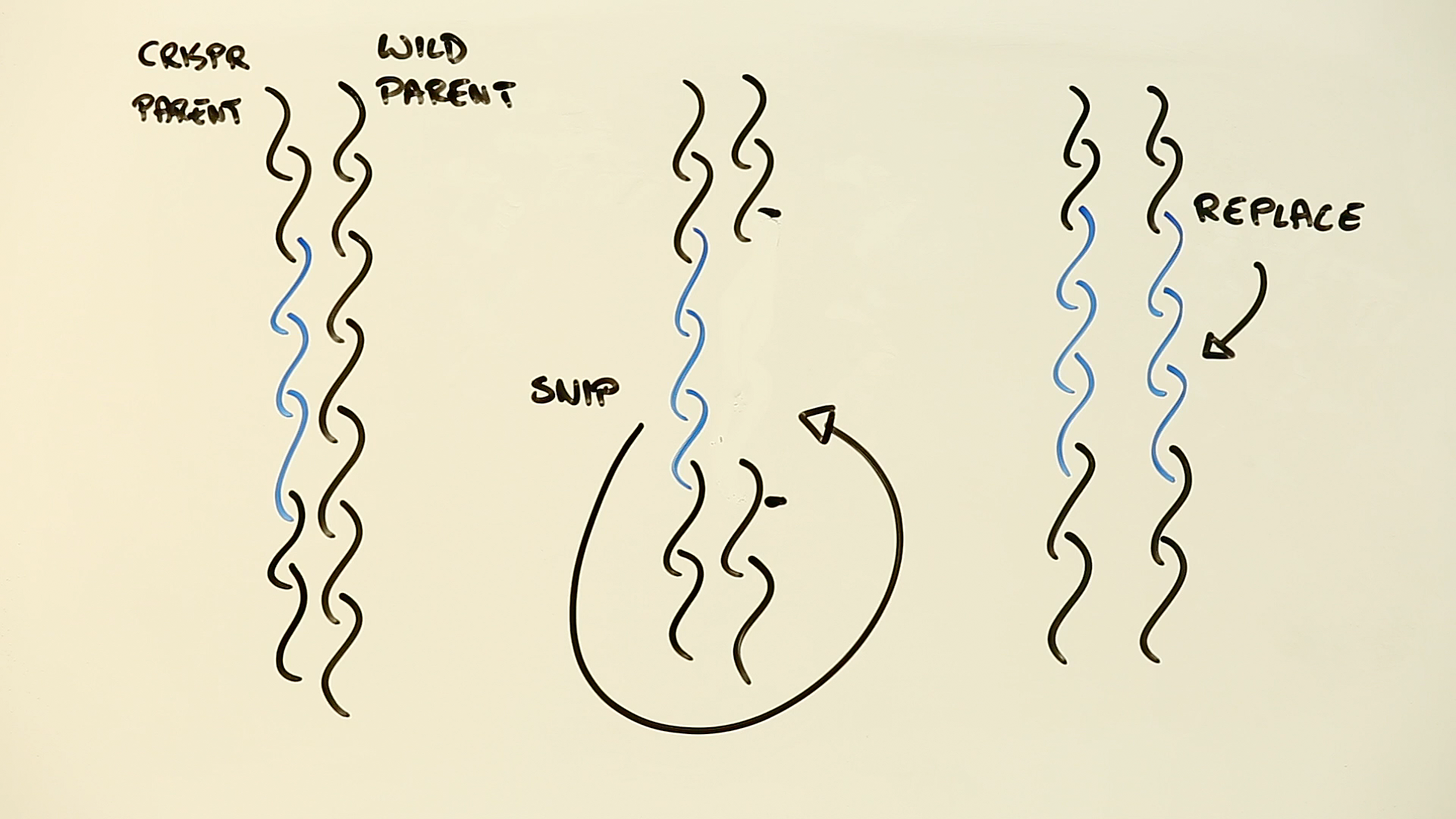
CRISPR uses specially designed molecules that run along the strands of DNA in an organism’s genome, and seek out specific sequences of genetic code. Once found, they snip out the old code, and paste in the new.
It’s essentially a sophisticated biological search and replace technology that allows scientists to easily modify the genetic traits of an organism – such as replacing the parts of a mosquito’s genome that allows it to host malaria-causing parasites for instance.
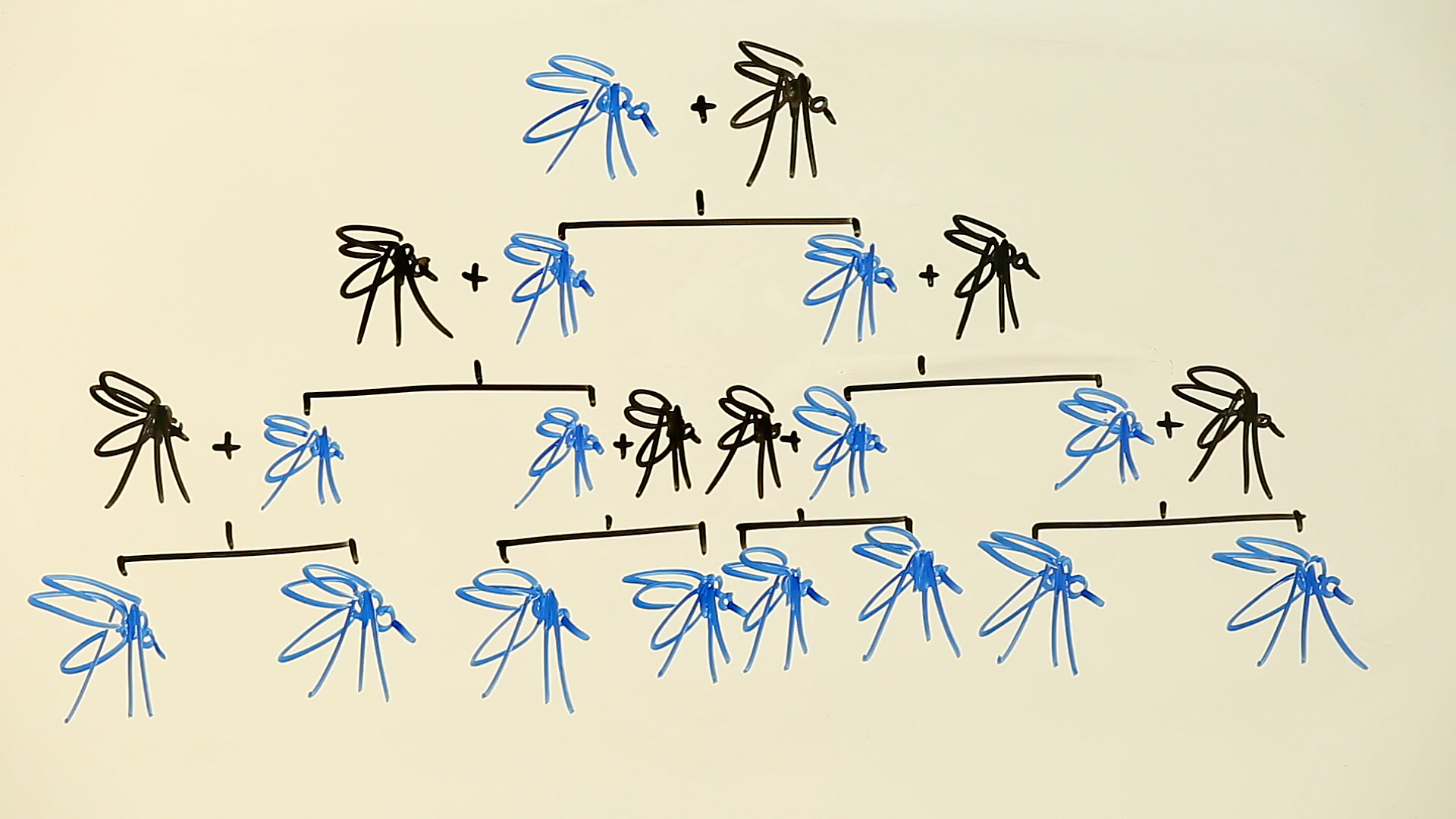
Unfortunately, on it’s own, CRISPR isn’t so effective. Every time a CRISPR mosquito mates with a wild one, its genetically modified DNA is diluted down, meaning that some of its offspring will still be able to carry the malaria parasite.
This is where the genius of CRISPR-enabled gene drives comes in. Imagine what would happen if that replaced sequence in the CRISPR mosquito also contained a bit of code that continued to search and replace parasite-enabling DNA sequences. When the mosquito mated, the built-in code would ensure that every single one of its progeny would inherit the same traits – as well as inheriting the CRISPR code that would ensure the anti-malaria gene was passed on to every future generation.
In other words, the new gene would be irresistibly driven though the whole mosquito population, and eventually, every mosquito would become a human designed malaria-free insect. And this isn’t a technology that is restricted to mosquitoes – any organism that reproduces by sharing DNA through sexual intercourse is fair game.
That said, designing an effective and responsible gene drive isn’t a piece of cake.
Working out which genes to change in an organism for instance is still an inexact science – especially when the CRISPR search and replace may modify more genetic sequences than expected, or introduce unanticipated traits.
The speed with which new genes can be driven through a species also depends on how frequently they reproduce. Gene drives are in principle great for transforming fast-breeding organisms like insects. Yet it would take millennia to push a gene drive through relatively a slow-breeding species like … humans, for instance. And that’s assuming that naturally occurring genetic mutations don’t mess up the re-coding in the meantime.
Nevertheless, gene drives do radically change how we’re able to manipulate nature. And that also means they raise tough questions around risk and responsibility.
For instance, we don’t yet know how gene drives might harmfully disrupt ecosystems, or create unexpected risks to other species – humans included. And we’re not sure how much hard wiring in certain genetic traits will weaken an organism’s ability to adapt to changing environments, especially when those traits would normally be rejected through natural selection.
And then there’s the danger of entrepreneurs, and hobbyists even, playing around with nature, just because they can. Or people who intentionally set out to create even more dangerous species – weaponized super-mosquitoes possibly, or hunger games-like species designed to maim and kill. Not to mention the possibility of groups starting exclusive human gene drives that will give their future heirs genetically enhanced abilities.
Because of concerns like these, scientists have already started to think about how gene drives might get out of hand. Recommendations are beginning to emerge on responsible practices, like testing gene drives in the lab before trying them out in the environment. And there’s talk of counter-measures such as follow-on gene drives that can reverse changes, or block the propagation of genetic modifications.
Yet these discussions often assume that it’s the scientists and the developers who know what’s best. I suspect though that, when it comes to intentionally redesigning natural species, there are a lot more people who would like a say in what should and should not happen. Especially as the risks and benefits of gene drives are likely to affect far more people than those who came up with the bright idea in the first place.
Which leaves the question: if the use of gene drives are a social issue as much as a scientific one, how can ordinary people become involved in ensuring we get it right before it’s too late?
(Watch the video here)